|
11/18/2023 0 Comments Carbon 14 dating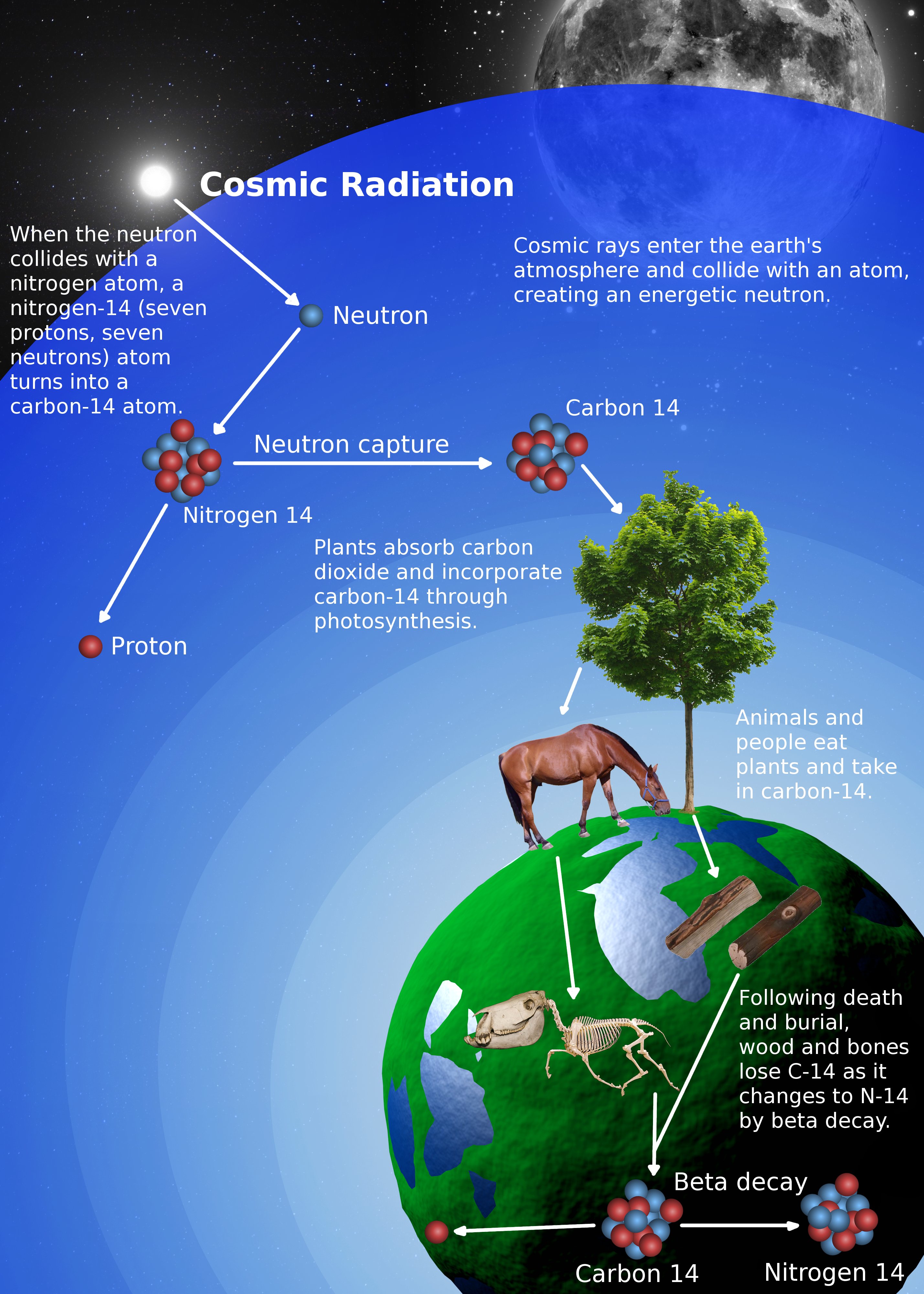 
From the discovery of Carbon-14 to radiocarbon dating of fossils, we can see what an essential role Carbon has played and continues to play in our lives today. Although it may be seen as outdated, many labs still use Libby's half-life in order to stay consistent in publications and calculations within the laboratory. We now use what is known as the Cambridge half-life of 5730+/- 40 years for Carbon-14. Carbon dating is used to work out the age of organic material in effect, any living thing. Throughout the years, measurement tools have become more technologically advanced, allowing researchers to be more precise. From that point on, scientists have used these techniques to examine fossils, rocks, and ocean currents as well as to determine age and event timing. The accuracy of this proposal was proven by dating a piece of wood from an Ancient Egyptian barge, the age of which was already known. Using this hypothesis, the initial half-life he determined was 5568, give or take 30 years. Using this finding, Willard Libby and his team at the University of Chicago proposed that Carbon-14 was unstable and underwent a total of 14 disintegrations per minute per gram. They found a form, an isotope, of Carbon that contained 8 neutrons and 6 protons. In 1940, Martin Kamen and Sam Ruben at the University of California, Berkeley Radiation Laboratory did just that. Before Radiocarbon dating was discovered, someone had to find the existence of the 14C isotope. He demonstrated the accuracy of radiocarbon dating by accurately estimating the age of wood from a series of samples for which the age was known, including an ancient Egyptian royal barge dating from 1850 BCE. In 1960, Libby was awarded the Nobel Prize in chemistry for this work. Libby estimated that the steady-state radioactivity concentration of exchangeable carbon-14 would be about 14 disintegrations per minute (dpm) per gram. Emilio Segrè asserted in his autobiography that Enrico Fermi suggested the concept to Libby at a seminar in Chicago that year. The technique of radiocarbon dating was developed by Willard Libby and his colleagues at the University of Chicago in 1949. Radiocarbon dating is therefore only useful for samples with ages of less than about 65,000 to 80,000 years.\). After several half-lives, too little 14C will remain in a sample for it to be useful for dating. This tells the scientists how long ago the organism died.ġ4C has a half-life of 5,730 years, meaning that after about five thousand years about half of the 14C will decay and turn into nitrogen. Scientists can study samples from the once-live creatures' remains to see how much radioactive 14C, as compared to the normal isotope of carbon (carbon-12 or 12C), is still around. The radioactive 14C gradually undergoes radioactive decay, transforming it into nitrogen, and therefore gradually "disappears". When the creature dies, it stops ingesting carbon. Some of the carbon is a radioactive isotope called carbon-14 ( 14C). Animals (and other heterotrophs) get their carbon by eating plants or other animals, from decaying organic matter, or from other similar sources. Plants (and other autotrophs) take in carbon dioxide gas from the atmosphere during photosynthesis. 
It is often used in archeology and some types of biology. 
Isotope Carbon-14 Dating (Radiocarbon Dating)Ĭarbon-14 dating (also called "radiocarbon dating") is used to determine the age of materials that contain carbon that was originally in living things.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
